- Anti-hestaminic & Respiratory Drugs (20)
- Anti-inflammatory Drugs (199) +-
- Baby & Mom (1349) +-
- Baby & Mom > Bath, skin & Hair > Skin Care > wibes (53)
- Beauty (3133) +-
- Beauty > Skin Care > whitening (308)
- Chemotherapy & Immune Response (891) +-
- Chemotherapy & Immune Response > ANTI-FUNGAL (11)
- Chemotherapy & Immune Response > Chemotherapeutic Agents > Hormone Antagonists >Enzyme Inhibitors (290)
- CIRCULATORY DISTURBANCE AGENTS (24)
- Diet & Fitness Products (284) +-
- DRUG AFFECTING CENTRAL NERVOUS SYSTEM (193)
- HEMATOLOGY (40)
-
Medical Supplies (510)
+-
- Chemicals & Disinfectants (19)
- Dental Supplies (31)
- Devices & Instruments (11)
- Diabetic Supplies (125)
- General Medical Supplies (21)
- I.V & Medical Solution (0)
- Intensive Care Unit & Anesthesia Supplies (0)
- KIDNEY UNIT SUPPLIES (21)
- Lab Supplies (3)
- Miscellaneous (21)
- Neonatal Unit Supplies (0)
- Operation Room Supplies (2)
- Sanitary (5)
- Sterilization Supplies (1)
- Surgical Sutures (4)
- Syringes (3)
-
Medicines & Health (2778)
+-
- Allergy & Sinus (98)
- Children's Health Care (54)
- Cough, Cold & Flu (289)
- Digestive Health & Nausea (243)
- Ear, Nose & Throat Care (187)
- Eye Care (134)
- Feminine Care (324)
- Foot Care (12)
- Orthopaedic Appliances (1)
- Pain Relief & Management (250)
- Pill Organizer (2)
- Skin Treatments (866)
- Sleep & Snoring Aids (2)
- Support & Braces (8)
- Medicines & health > Gout releif (40)
- Natural & Organic Products (81) +-
- OTC > Analgesics > Anti-inflammatory Drugs (47)
-
Personal Care (3387)
+-
- Bath & Body (273)
- Deodorant & Anti-perspirants (191)
- Ear, Nose & Throat Care (183)
- Eye Care (137)
- Feminine Care (374)
- Foot Care (20)
- Hair Care (513)
- Home Tests & Monitorings (17)
- Incontinence (7)
- Lip Care (26)
- Massage & Relaxation (17)
- Natural & Organic Personal Care (7)
- Oral Care (91)
- Pregnancy & Fertility (66)
- Shaving & Grooming (75)
- Sun Care (81)
-
Prescription Drugs (2992)
+-
- Analgesics (186)
- Cardiovascular System (383)
- Drugs Affecting Musculoskeletal System (65)
- Drugs Used In Infections (56)
- Ear & Nose Drugs (2)
- Endocrine System (190)
- Gastrointestinal Tract (255)
- Gastrointestinal Tract > Hepatology > Liver treatment (61)
- GYNECOLOGY (2)
- Miscellaneous (11)
- NEPHROLOGY > URINARY SYSTEM > RENAL DISORDERS > URINARY TRACT DISORDERS (47)
- NEUROLOGY (234)
- Nutrients & Blood Electrolytes (2)
- Respiratory System (157)
- SKIN > NAILS > HAIR > TOPICAL PREPARATIONS (115)
- Vaccines (1)
- Prescription drugs > Cardiovascular system > Anti-hypertension drugs (248)
- Sexual Wellness (315) +-
- Vitamins & Minerals Supplements (1248) +-
Ex Tax: 22EGP
Example
You can return the product within 14 days of purchase.
ReturnsYou can return the product within 14 days of purchase.

Prescription Information
Composition:
Each film coated tablet contains:
- Silden 25 mg Film coated Tablets:
Sildenafil citrate …………………….…………….…….…….. 35.12 mg
Equivalent to Sildenafil …………………………...……………. 25 mg
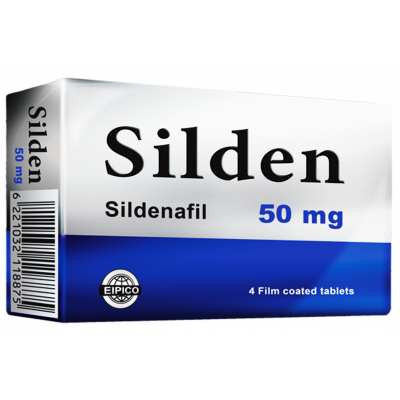
- Silden 50 mg Film coated Tablets:
Sildenafil citrate ……………………………………….…………. 70 mg
Equivalent to Sildenafil …………………………………….…. 50 mg
- Silden 100 mg Film coated Tablets:
Sildenafil citrate …………………………………….………..…. 140 mg
Equivalent to Sildenafil …………………………………….... 100 mg
Inactive ingredients:
Lactose monohydrate, microcrystalline cellulose PH 102, croscarmellose sodium, magnesium stearate, colloidal silicon dioxide (Aerosil 200), purified talc, hydroxypropyl methylcellulose 2910, hydroxypropyl cellulose LF, dibutyl phthalate, titanium dioxide, patent blue, sicko indigotine lake )E132(, methylene chloride, methanol.
Povidone K 30 for 50 and 100 mg Film coated Tablets.
Therapeutic Indications
Silden is indicated in adult men with erectile dysfunction, which is the inability to achieve or maintain a penile erection sufficient for satisfactory sexual performance.
In order for Silden to be effective, sexual stimulation is required.
Dosage and Administration
Dosage:
Use in Adults:
The recommended dose is 50 mg taken as needed approximately one hour before sexual activity. Based on efficacy and tolerability, the dose may be increased to 100 mg or decreased to 25 mg. The maximum recommended dose is 100 mg.
The maximum recommended dosing frequency is once per day.
If Silden is taken with food, the onset of activity may be delayed compared to the fasted state.
Special populations:
Elderly:
Dosage adjustments are not required in elderly patients (≥ 65 years old).
Renal impairment:
The dosing recommendations described in “Use in Adults” apply to patients with mild to moderate renal impairment (creatinine clearance = 30-80 mL/min).
Since sildenafil clearance is reduced in patients with severe renal impairment (creatinine clearance < 30 mL/min), a 25 mg dose should be considered. Based on efficacy and tolerability, the dose may be increased stepwise to 50 mg up to 100 mg as necessary.
Hepatic impairment:
Since sildenafil clearance is reduced in patients with hepatic impairment (e.g. cirrhosis), a 25 mg dose should be considered. Based on efficacy and tolerability, the dose may be increased stepwise to 50 mg up to 100 mg as necessary.
Pediatric population:
Silden is not indicated for individuals below 18 years of age.
Use in patients taking other medicinal products:
With the exception of ritonavir for which co-administration with Silden is not advised, a starting dose of 25 mg should be considered in patients receiving concomitant treatment with CYP3A4 inhibitors.
In order to minimize the potential of developing postural hypotension in patients receiving alpha-blocker treatment, patients should be stabilized on alpha-blocker therapy prior to initiating Silden treatment. In addition, initiation of Silden at a dose of 25 mg should be considered.
Administration:
For oral use.
Contraindications:
Hypersensitivity to the active substance or to any of the excipients.
Consistent with its known effects on the nitric oxide/cyclic guanosine monophosphate (cGMP) pathway, sildenafil was shown to potentiate the hypotensive effects of nitrates, and its co-administration with nitric oxide donors (such as amyl nitrite) or nitrates in any form is therefore contraindicated.
The co-administration of PDE5 inhibitors, including Silden, with guanylate cyclase stimulators, such as riociguat, is contraindicated as it may potentially lead to symptomatic hypotension.
Agents for the treatment of erectile dysfunction, including Silden, should not be used in men for whom sexual activity is inadvisable (e.g. patients with severe cardiovascular disorders such as unstable angina or severe cardiac failure).
Silden is contraindicated in patients who have loss of vision in one eye because of nonarteritic anterior ischemic optic neuropathy (NAION), regardless of whether this episode was in connection or not with previous PDE5 inhibitor exposure.
The safety of sildenafil has not been studied in the following subgroups of patients and its use is therefore contraindicated: severe hepatic impairment, hypotension (blood pressure < 90/50 mmHg), recent history of stroke or myocardial infarction and known hereditary degenerative retinal disorders such as retinitis pigmentosa (a minority of these patients have genetic disorders of retinal phosphodiesterases).
Warnings and Precautions:
A medical history and physical examination should be undertaken to diagnose erectile dysfunction and determine potential underlying causes, before pharmacological treatment is considered.
Cardiovascular risk factors:
Prior to initiating any treatment for erectile dysfunction, physicians should consider the cardiovascular status of their patients, since there is a degree of cardiac risk associated with sexual activity. Sildenafil has vasodilator properties, resulting in mild and transient decreases in blood pressure. Prior to prescribing Silden, physicians should carefully consider whether their patients with certain underlying conditions could be adversely affected by such vasodilatory effects, especially in combination with sexual activity. Patients with increased susceptibility to vasodilators include those with left ventricular outflow obstruction (e.g. aortic stenosis, hypertrophic obstructive cardiomyopathy), or those with the rare syndrome of multiple system atrophy manifesting, as severely impaired autonomic control of blood pressure.
Sildenafil potentiates the hypotensive effect of nitrates.
Serious cardiovascular events, including myocardial infarction, unstable angina, sudden cardiac death, ventricular arrhythmia, cerebrovascular hemorrhage, transient ischemic attack, hypertension and hypotension have been reported post marketing in temporal association with the use of sildenafil. Most, but not all, of these patients had pre-existing cardiovascular risk factors. Many events were reported to occur during or shortly after sexual intercourse and a few were reported to occur shortly after the use of sildenafil without sexual activity. It is not possible to determine whether these events are related directly to these factors or to other factors.
Priapism:
Agents for the treatment of erectile dysfunction, including Silden, should be used with caution in patients with anatomical deformation of the penis (such as angulation, cavernosal fibrosis or Peyronie's disease), or in patients who have conditions, which may predispose them to priapism (such as sickle cell anemia, multiple myeloma or leukemia).
Prolonged erections and priapism have been reported with sildenafil in post-marketing experience. In the event of an erection that persists for longer than 4 hours, the patient should seek immediate medical assistance. If priapism is not treated immediately, penile tissue damage and permanent loss of potency could result.
Concomitant use with other PDE5 inhibitors or other treatments for erectile dysfunction:
The safety and efficacy of combinations of sildenafil with other PDE5 inhibitors, or other pulmonary arterial hypertension (PAH) treatments containing sildenafil, or other treatments for erectile dysfunction have not been studied.
Therefore, the use of such combinations is not recommended.
Effects on vision:
Cases of visual defects have been reported spontaneously in connection with the intake of sildenafil and other PDE5 inhibitors. Cases of nonarteritic anterior ischemic optic neuropathy, a rare condition, have been reported spontaneously and in an observational study in connection with the intake of sildenafil and other PDE5 inhibitors. Patients should be advised that in the event of any sudden visual defect, they should stop taking Silden and consult a physician immediately.
Concomitant use with ritonavir:
Co-administration of Silden with ritonavir is not advised.
Concomitant use with alpha-blockers:
Caution is advised when Silden is administered to patients taking an alpha-blocker, as the co-administration may lead to symptomatic hypotension in a few susceptible individuals. This is most likely to occur within 4 hours post Silden dosing. In order to minimise the potential for developing postural hypotension, patients should be hemodynamically stable on alpha-blocker therapy prior to initiating Silden treatment. Initiation of Silden at a dose of 25 mg should be considered. In addition, physicians should advise patients what to do in the event of postural hypotensive symptoms.
Effect on bleeding:
Studies with human platelets indicate that sildenafil potentiates the antiaggregatory effect of sodium nitroprusside in vitro.
There is no safety information on the administration of sildenafil to patients with bleeding disorders or active peptic ulceration. Therefore, Silden should be administered to these patients only after careful benefit risk assessment.
The film coating of the tablet contains lactose. Silden should not be administered to men with rare hereditary problems of galactose intolerance, Lapp lactase deficiency or glucosegalactose malabsorption.
Women:
Silden is not indicated for use by women.
Drug Interactions
Effects of other Medicinal products on Silden:
In vitro studies:
Sildenafil metabolism is principally mediated by the cytochrome P450 (CYP) isoforms 3A4 (major route) and 2C9 (minor route). Therefore, inhibitors of these isoenzymes may reduce sildenafil clearance and inducers of these isoenzymes may increase sildenafil clearance.
In vivo studies:
Population pharmacokinetic analysis of clinical trial data indicated a reduction in sildenafil clearance when co-administered with CYP3A4 inhibitors (such as ketoconazole, erythromycin, cimetidine). Although no increased incidence of adverse events was observed in these patients, when Silden is administered concomitantly with CYP3A4 inhibitors, a starting dose of 25 mg should be considered.
Co-administration of the HIV protease inhibitor ritonavir, which is a highly potent P450 inhibitor, at steady state (500 mg twice daily) with sildenafil (100 mg single dose) resulted in a 300% (4 fold) increase in sildenafil Cmaxand a 1,000% (11 fold) increase in sildenafil plasma AUC. At 24 hours, the plasma levels of sildenafil were still approximately 200 ng/mL, compared to approximately 5 ng/mL when sildenafil was administered alone. This is consistent with ritonavir's marked effects on a broad range of P450 substrates. Sildenafil had no effect on ritonavir pharmacokinetics. Based on these pharmacokinetic results, co-administration of Silden with ritonavir is not advised and in any event the maximum dose of Silden should under no circumstances exceed 25 mg within 48 hours.
Co-administration of the HIV protease inhibitor saquinavir, a CYP3A4 inhibitor, at steady state (1200 mg three times a day) with sildenafil (100 mg single dose) resulted in a 140% increase in sildenafil Cmaxand a 210% increase in sildenafil AUC. Sildenafil had no effect on saquinavir pharmacokinetics. Stronger CYP3A4 inhibitors such as ketoconazole and itraconazole would be expected to have greater effects.
When a single 100 mg dose of sildenafil was administered with erythromycin, a moderate CYP3A4 inhibitor, at steady state (500 mg twice daily for 5 days), there was a 182% increase in sildenafil systemic exposure (AUC). In normal healthy male volunteers, there was no evidence of an effect of azithromycin (500 mg daily for 3 days) on the AUC, Cmax, tmax, elimination rate constant, or subsequent half-life of sildenafil or its principal circulating metabolite. Cimetidine (800 mg), a cytochrome P450 inhibitor and nonspecific CYP3A4 inhibitor, caused a 56% increase in plasma sildenafil concentrations when co-administered with sildenafil (50 mg) to healthy volunteers.
Grapefruit juice is a weak inhibitor of CYP3A4 gut wall metabolism and may give rise to modest increases in plasma levels of sildenafil.
Single doses of antacid (magnesium hydroxide/aluminum hydroxide) did not affect the bioavailability of sildenafil.
Although specific interaction studies were not conducted for all medicinal products, population pharmacokinetic analysis showed no effect of concomitant treatment on sildenafil pharmacokinetics when grouped as CYP2C9 inhibitors (such as tolbutamide, warfarin, phenytoin), CYP2D6 inhibitors (such as selective serotonin reuptake inhibitors, tricyclic antidepressants), thiazide and related diuretics, loop and potassium sparing diuretics, angiotensin converting enzyme inhibitors, calcium channel blockers, beta-adrenoreceptor antagonists or inducers of CYP450 metabolism (such as rifampicin, barbiturates). In a study of healthy male volunteers, co-administration of the endothelin-receptor antagonist, bosentan, (an inducer of CYP3A4 [moderate], CYP2C9 and possibly of CYP2C19) at steady state (125 mg twice a day) with sildenafil at steady state (80 mg three times a day) resulted in 62.6% and 55.4% decrease in sildenafil AUC and Cmax, respectively. Therefore, concomitant administration of strong CYP3A4 inducers, such as rifampin, is expected to cause
greater decreases in plasma concentrations of Silden.
Nicorandil is a hybrid of potassium channel activator and nitrate. Due to the nitrate component, it has the potential to result in a serious interaction with Silden.
Effects of Silden on other Medicinal products:
In vitro studies:
Sildenafil is a weak inhibitor of the cytochrome P450 isoforms 1A2, 2C9, 2C19, 2D6, 2E1 and 3A4 (IC50>150 μM).
Given sildenafil peak plasma concentrations of approximately 1 μM after recommended doses, it is unlikely that sildenafil will alter the clearance of substrates of these isoenzymes.
There are no data on the interaction of sildenafil and nonspecific phosphodiesterase inhibitors such as theophylline or dipyridamole.
In vivo studies:
Consistent with its known effects on the nitric oxide/cGMP pathway, sildenafil was shown to potentiate the hypotensive effects of nitrates, and its co-administration with nitric oxide donors or nitrates in any form is therefore contraindicated.
There was no evidence of favorable clinical effect of the combination of sildenafil and riociguat in the population studied. Concomitant use of riociguat with PDE5 inhibitors, including sildenafil, is contraindicated.
Concomitant administration of sildenafil to patients taking alpha-blocker therapy may lead to symptomatic hypotension in a few susceptible individuals. This is most likely to occur within 4 hours post sildenafil dosing.
In three specific drug-drug interaction studies, the alpha-blocker doxazosin (4 mg and 8 mg) and sildenafil (25 mg, 50 mg, or 100 mg) were administered simultaneously to patients with benign prostatic hyperplasia (BPH) stabilized on doxazosin therapy. In these study populations, mean additional reductions of supine blood pressure of 7/7 mmHg, 9/5 mmHg, and 8/4 mmHg, and mean additional reductions of standing blood pressure of 6/6 mmHg, 11/4 mmHg, and 4/5 mmHg, respectively, were observed. When sildenafil and doxazosin were administered simultaneously to patients stabilized on doxazosin therapy, there were infrequent reports of patients who experienced symptomatic postural hypotension. These reports included dizziness and lightheadedness, but not syncope.
No significant interactions were shown when sildenafil (50 mg) was co-administered with tolbutamide (250 mg) or warfarin (40 mg), both of which are metabolized by CYP2C9.
Sildenafil (50 mg) did not potentiate the increase in bleeding time caused by acetyl salicylic acid (150 mg).
Sildenafil (50 mg) did not potentiate the hypotensive effects of alcohol in healthy volunteers with mean maximum blood alcohol levels of 80 mg/dl.
Pooling of the following classes of antihypertensive medication; diuretics, beta blockers, ACE inhibitors, angiotensin II antagonists, antihypertensive medicinal products (vasodilator and centrally acting), adrenergic neuron blockers, calcium channel blockers and alpha adrenoceptor blockers, showed no difference in the side effect profile in patients taking sildenafil compared to placebo treatment. In a specific interaction study, where sildenafil (100 mg) was co-administered with amlodipine in hypertensive patients, there was an additional reduction on supine systolic blood pressure of 8 mmHg. The corresponding additional reduction in supine diastolic blood pressure was 7 mmHg. These additional blood pressure reductions were of a similar magnitude to those seen when sildenafil was administered alone to healthy volunteers.
Sildenafil (100 mg) did not affect the steady state pharmacokinetics of the HIV protease inhibitors, saquinavir and ritonavir, both of which are CYP3A4 substrates.
In healthy male volunteers, sildenafil at steady state (80 mg t.i.d.) resulted in a 49.8% increase in bosentan AUC and a 42% increase in bosentan Cmax(125 mg b.i.d.).
Pregnancy and Lactation
Silden is not indicated for use by women.
There are no adequate and well controlled studies in pregnant or breastfeeding women.
There was no effect on sperm motility or morphology after single 100 mg oral doses of sildenafil in healthy volunteers.
Effects on ability to drive and to use machines
Silden may have a minor influence on the ability to drive and to use machines.
As dizziness and altered vision were reported in clinical trials with sildenafil, patients should be aware of how they react to Silden, before driving or operating machinery.
Undesirable Effects:
Adverse reactions from post marketing:
Surveillance has been gathered covering an estimated period >10 years.
Because not all adverse reactions are reported and included in the safety database, the frequencies of these reactions cannot be reliably determined.
List of adverse reactions:
In the list below all medically important adverse reactions, which occurred in clinical trials at an incidence greater than placebo are listed by system organ class and frequency (Very common (≥1/10); Common (≥1/100 to <1/10); Uncommon
(≥1/1,000 to <1/100); Rare (≥1/10,000 to <1/1,000).
Within each frequency grouping, undesirable effects are presented in order of decreasing seriousness.
Medically Important Adverse Reactions Reported At An Incidence Greater Than Placebo In Controlled Clinical Studies And Medically Important Adverse Reactions Reported Through Post marketing Surveillance:
Infections and Infestations:
Uncommon: Rhinitis.
Immune system disorders:
Uncommon: Hypersensitivity.
Nervous system disorders:
Very common: Headache.
Common: Dizziness.
Uncommon: Somnolence, hypoaesthesia.
Rare: Cerebrovascular accident, transient ischemic attack, seizure*, seizure recurrence*, syncope.
Eye disorders:
Common: Visual color distortions**, visual disturbance, blurred vision.
Uncommon: Lacrimation disorders***, eye pain, photophobia, photopsia, ocular hyperaemia, visual brightness, conjunctivitis.
Rare: Non-arteritic anterior ischemic optic neuropathy (NAION)*, retinal vascular occlusion*, retinal haemorrhage, arteriosclerotic retinopathy, retinal disorder, glaucoma, visual field defect, diplopia, reduced visual acuity, myopia, asthenopia, vitreous floaters, iris disorder, mydriasis, halo vision, eye oedema, eye swelling, eye disorder, conjunctival hyperaemia, eye irritation, abnormal sensation in eye, eyelid oedema, scleral discoloration.
Ear and Labyrinth disorders:
Uncommon: Vertigo, tinnitus.
Rare: Deafness.
Cardiac disorders:
Uncommon: Tachycardia, palpitations.
Rare: Sudden cardiac death*, myocardial infarction, ventricular arrhythmia*, atrial fibrillation, unstable angina.
Vascular disorders:
Common: Flushing, hot flush.
Uncommon: Hypertension, hypotension.
Respiratory, Thoracic and Mediastinal disorders:
Common: Nasal congestion.
Uncommon: Epistaxis, sinus congestion.
Rare: Throat tightness, nasal oedema, nasal dryness.
Gastrointestinal disorders:
Common: Nausea, dyspepsia.
Uncommon: Gastroesophageal reflux disease, vomiting, upper abdominal pain, dry mouth.
Rare: Oral hypoaesthesia.
Skin and Subcutaneous tissue disorders:
Uncommon: Rash.
Rare: Stevens-Johnson Syndrome (SJS)*, Toxic Epidermal Necrolysis (TEN)*.
Musculoskeletal and Connective tissue disorders:
Uncommon: Myalgia, pain in extremity.
Renal and Urinary disorders:
Uncommon: Haematuria.
Reproductive system and Breast disorders:
Rare: Penile haemorrhage, priapism*, haematospermia, increased erection.
General disorders and Administration site conditions:
Uncommon: Chest pain, fatigue, feeling hot.
Rare: Irritability.
Investigations:
Uncommon: Increased heart rate.
* Reported during post marketing surveillance only.
** Visual colour distortions: Chloropsia, chromatopsia, cyanopsia, erythropsia and xanthopsia.
*** Lacrimation disorders: Dry eye, lacrimal disorder and increased lacrimation.
Overdosage
In single dose volunteer studies of doses up to 800 mg, adverse reactions were similar to those seen at lower doses, but the incidence rates and severities were increased. Doses of 200 mg did not result in increased efficacy but the incidence of adverse reactions (headache, flushing, dizziness, dyspepsia, nasal congestion, altered vision) was increased.
In cases of overdose, standard supportive measures should be adopted as required. Renal dialysis is not expected to accelerate clearance, as sildenafil is highly bound to plasma proteins and not eliminated in the urine.
Pharmacodynamic properties:
Pharmacotherapeutic group: Urologicals; Drugs used in erectile dysfunction.
Mechanism of action:
Silden is an oral therapy for erectile dysfunction. In the natural setting, i.e. with sexual stimulation, it restores impaired erectile function by increasing blood flow to the penis.
The physiological mechanism responsible for erection of the penis involves the release of nitric oxide (NO) in the corpus cavernosum during sexual stimulation. Nitric oxide then activates the enzyme guanylate cyclase, which results in increased levels of cyclic guanosine monophosphate (cGMP), producing smooth muscle relaxation in the corpus cavernosum and allowing inflow of blood.
Sildenafil is a potent and selective inhibitor of cGMP specific phosphodiesterase type 5 (PDE5) in the corpus cavernosum, where PDE5 is responsible for degradation of cGMP. Sildenafil has a peripheral site of action on erections.
Sildenafil has no direct relaxant effect on isolated human corpus cavernosum but potently enhances the relaxant effect of NO on this tissue. When the NO/cGMP pathway is activated, as occurs with sexual stimulation, inhibition of PDE5 by sildenafil results in increased corpus cavernosum levels of cGMP. Therefore, sexual stimulation is required in order for sildenafil to produce its intended beneficial pharmacological effects.
Pharmacodynamic effects:
Studies in vitro have shown that sildenafil is selective for PDE5, which is involved in the erection process. Its effect is more potent on PDE5 than on other known phosphodiesterases. There is a 10-fold selectivity over PDE6, which is involved in the phototransduction pathway in the retina. At maximum recommended doses, there is an 80 fold selectivity over PDE1, and over 700 fold over PDE 2, 3, 4, 7, 8, 9, 10 and 11. In particular, sildenafil has greater than 4,000 fold selectivity for PDE5 over PDE3, the cAMP specific phosphodiesterase isoform involved in the control of cardiac contractility.
Pharmacokinetic properties:
Absorption:
Sildenafil is rapidly absorbed. Maximum observed plasma concentrations are reached within 30 to 120 minutes (median 60 minutes) of oral dosing in the fasted state. The mean absolute oral bioavailability is 41% (range 25 - 63%).
After oral dosing of sildenafil, AUC and Cmaxincrease in proportion with dose over the recommended dose range (25-100 mg).
When sildenafil is taken with food, the rate of absorption is reduced with a mean delay in tmaxof 60 minutes and a mean reduction in Cmaxof 29%.
Distribution:
The mean steady state volume of distribution (Vd) for sildenafil is 105 L, indicating distribution into the tissues. After a single oral dose of 100 mg, the mean maximum total plasma concentration of sildenafil is approximately 440 ng/mL (CV 40%). Since sildenafil (and its major circulating N-desmethyl metabolite) is 96% bound to plasma proteins, this results in the mean maximum free plasma concentration for sildenafil of 18 ng/mL (38 nM). Protein binding is independent of total drug concentrations.
In healthy volunteers receiving sildenafil (100 mg single dose), less than 0.0002% (average 188 ng) of the administered dose was present in ejaculate 90 minutes after dosing.
Biotransformation:
Sildenafil is cleared predominantly by the CYP3A4 (major route) and CYP2C9 (minor route) hepatic microsomal isoenzymes. The major circulating metabolite results from N-demethylation of sildenafil. This metabolite has a phosphodiesterase selectivity profile similar to sildenafil and an in vitro potency for PDE5 approximately 50% that of the parent drug. Plasma concentrations of this metabolite are approximately 40% of those seen for sildenafil. The N-desmethyl metabolite is further metabolized, with a terminal half-life of approximately 4 h.
Elimination:
The total body clearance of sildenafil is 41 L/h with a resultant terminal phase half-life of 35h. After either oral or intravenous administration, sildenafil is excreted as metabolites, predominantly in the feces (approximately 80% of administered oral dose) and to a lesser extent in the urine (approximately 13% of administered oral dose).
Pharmacokinetics in Special patient groups:
Elderly:
Healthy elderly volunteers (65 years or over) had a reduced clearance of sildenafil, resulting in approximately 90% higher plasma concentrations of sildenafil and the active N-desmethyl metabolite compared to those seen in healthy younger volunteers (18-45 years). Due to age differences in plasma protein binding, the corresponding increase in free sildenafil plasma concentration was approximately 40%.
Renal insufficiency:
In volunteers with mild to moderate renal impairment (Creatinine Clearance = 30-80 mL/min), the pharmacokinetics of sildenafil were not altered after receiving a 50 mg single oral dose. The mean AUC and Cmaxof the N-desmethyl metabolite increased up to 126% and up to 73% respectively, compared to age matched volunteers with no renal impairment. However, due to high inter-subject variability, these differences were not statistically significant. In volunteers with severe renal impairment (Creatinine Clearance < 30 mL/min), sildenafil clearance was reduced, resulting in mean increases in AUC and Cmaxof 100% and 88% respectively, compared to age matched volunteers with no renal impairment. In addition, N-desmethyl metabolite AUC and Cmaxvalues were significantly increased by 200% and 79% respectively.
Hepatic insufficiency:
In volunteers with mild to moderate hepatic cirrhosis (Child Pugh A and B), sildenafil clearance was reduced, resulting in increases in AUC (84%) and Cmax(47%) compared to age matched volunteers with no hepatic impairment. The pharmacokinetics of sildenafil in patients with severely impaired hepatic function have not been studied.
Storage
Store in a dry place at a temperature not exceeding 30°C.
Packaging
Silden 25 mg Film coated Tablets: Box containing 1 strip of 10 film coated tablets.
Silden 50 mg Film coated Tablets: Box containing 1 strip of 4 film coated tablets.
Silden 100 mg Film coated Tablets: Box containing 1 strip of 4 film coated tablets.

Write a review
Your Name:Your Review: Note: HTML is not translated!
Rating: Bad Good
Enter the code in the box below: